The endoplasmic reticulum (ER) is a highly dynamic organelle that undergoes significant morphological alterations in response to cellular stress. While conventional transmission electron microscopy (TEM) has provided valuable insights into these changes, such as the formation of crystalloid-ER and ER whorls, obtaining comprehensive three-dimensional (3D) information on these large structures within their cellular context has remained a challenge. To overcome these limitations, this study introduces an innovative application of dual-axis scanning transmission electron microscopy (STEM) tomography to investigate ER morphology under stress conditions in human embryonic kidney (HEK) cells overexpressing the cation channel polycystin-2 (PC-2). Benefitting from high-resolution, increased depth-of-focus, and reduced aberrations, STEM tomography enabled the detailed 3D reconstruction of large cellular subvolumes, providing unprecedented views of stress-induced ER structures. Our findings reveal distinct ultrastructural details of both crystalloid-ER and ER whorls. Crystalloid-ER exhibited a tubular architecture with potential interconnectedness, while ER whorls displayed a lamellar organisation and distinct membrane curvature. We observed the co-occurrence of these distinct smooth ER (sER) morphotypes within the same cell, yet they remained spatially separated, suggesting potential functional specialisation. Furthermore, we identified direct membrane contacts in mixed morphotypes, hinting at a shared origin or dynamic relationship between these structures. The study also elucidated the interactions of these organised smooth ER (OSER) structures with other organelles, such as mitochondria (MAM sites) and vesicles. In summary, the presented ultra-structural insights have a significant impact on our understanding of stress-related ER morphology changes. The ability to visualise the intricate 3D architecture and spatial relationships of these structures provides novel perspectives on the ER’s adaptive responses to stress, including potential roles in lipid and protein biosynthesis and intracellular communication. These findings underscore the power of dual-axis STEM tomography for elucidating complex organellar organisation and dynamics in their native cellular context.
Heinz, V., Rachel, R., & Ziegler, C. (2025). Application of STEM tomography to investigate smooth ER morphology under stress conditions. Journal of Microscopy, 299(3), 228-241. https://doi.org/10.1111/jmi.70020
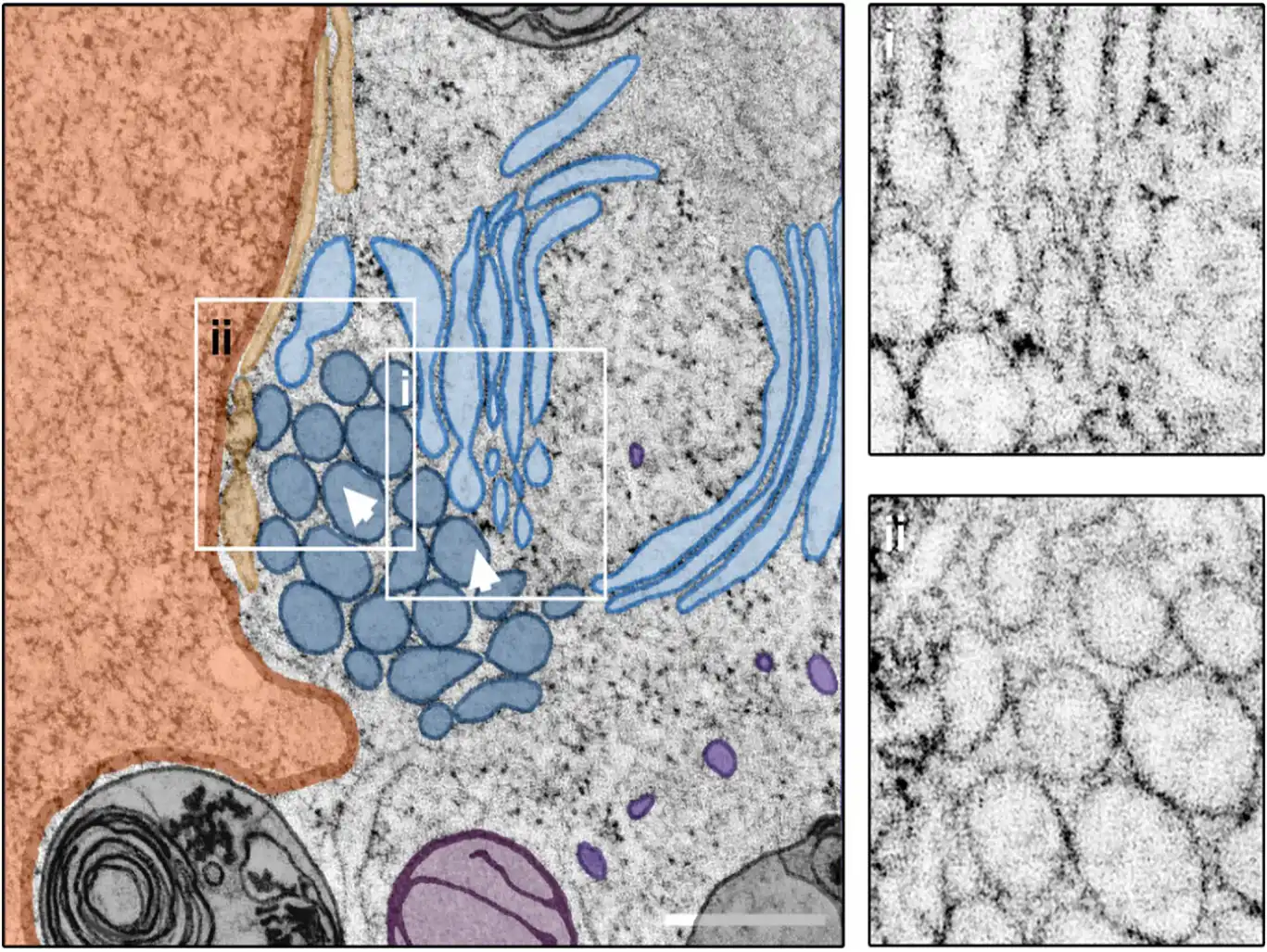
Image: Mixed crystalloid-ER and whorl morphology. Central slice through a STEM tomogram showing a patch of crystalloid-ER (dark blue) located in close proximity to a putative ER whorl (light blue) in its intracellular environment. Closer inspection of the reconstructed tomogram reveals a direct connection between the two ER morphotypes (I), as well as the presence of multiple vesicles (not present in the central tomogram section), located near the nucleus (orange) and contacting the crystalloid ER (II). Arrowheads indicate the viewing direction in Figure 6. Scale bar = 500 nm. Credit: Journal of Microscopy